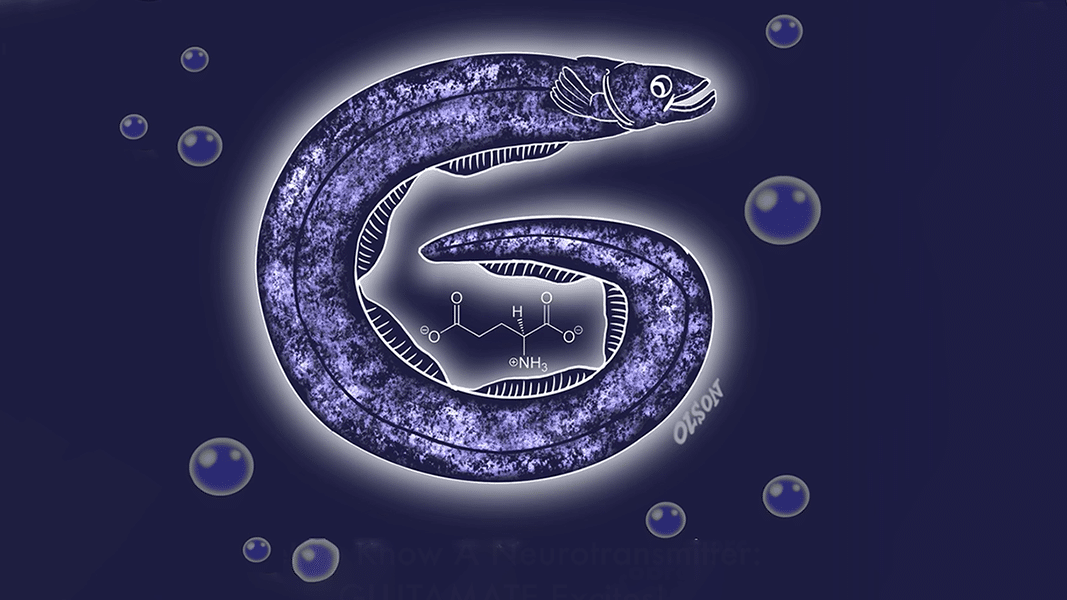
Glutamate, how do I love you? Let me count the ways … You power my brain. You dispose of my nitrogen. You trigger my nerves. You grow my synapses. You help me learn. You store my memories. And when I take ketamine, your absence transports me to bliss.
We could go on in the style of Elizabeth Barrett Browning – but the most important fact about glutamate is simple: it’s the principal excitatory neurotransmitter in the brain.
When a synapse wants to fire, when a neuron wants to signal, glutamate is the green light. It’s how the brain says “yes.”
In opposition is the neurotransmitter GABA, the most common inhibitory neurotransmitter. GABA is actually synthesized from glutamate, and both are regulated by the endocannabinoid system (ECS ). This speaks to the modulatory power of cannabinoids and their control over both excitation and inhibition.
And in the middle? Balance.
A Building Block of Proteins
Glutamate is not only a neurotransmitter, it’s also an amino acid – a building block of proteins. Indeed, it’s the most common amino acid in our diet and gives rise to the flavor known as umami. This ubiquity is part of the reason that it took so long for glutamate to be identified as a vital neurotransmitter. Only in the 1970s did the concept of glutamate as a neurotransmitter see widespread support, decades after the identification of acetylcholine, norepinephrine, serotonin, dopamine, and other messenger molecules. Apparently, it was hard for scientists to fathom that this common food ingredient powered the brain’s electrical system.
The blood-brain barrier – the protective system that keeps your blood from flooding your brain – does an excellent job at blocking out glutamate because the glutamate levels found in your blood would be quite toxic to your neurons. With that protection in place, eating too much glutamate does not appear to harm your brain. However, in situations like epilepsy, stroke or traumatic brain injury, the resulting surge of glutamate can cause brain damage via overexcitation (excitotoxicity). In those cases, pharmaceutical drugs that block glutamate from its receptors can prevent long-term damage.
Glutamate controls the excitability of neurons from outside the cell. Because the glutamate receptors are on the cell’s exterior, it’s the levels of glutamate in the extracellular fluid that matter. To regulate those levels, cells reduce glutamate by pumping it back into themselves across the membrane. Since glutamate is the major mediator of neuronal excitation as well as nervous system plasticity (learning), the importance of having the right concentrations in the right locations at the right times is paramount. One theory of schizophrenia is that it’s a dysfunction of the glutamate system.
The machinery of the glutamate system is so widespread and complex that it’s beyond the scope of this short review. Besides the many different transporters, receptors, and enzymes that regulate glutamate levels, glutamate itself can occur in a number of different shapes (conformations) that alter its function and what enzymes bind to it. Glutamate’s centrality to the nervous system makes it a key nexus for a huge number of diseases. And the ability of the endocannabinoid system to regulate the ebbs and flows of glutamate is a key reason that cannabinoids can help such a remarkably wide range of disorders.
How It Works

In the brain, there exists neurons that primarily release glutamate. These glutamergic neurons reside all across the brain, and on their surface they have CB1 cannabinoid receptors. When activated the CB1 receptors cause the release of glutamate in that area and the strengthening of neural signaling. The endocannabinoids strengthen synaptic transmission by stimulating the astrocytes (regulatory immune cells of the brain) via glutamate. This is one of the ways that the endocannabinoid system utilizes glutamate in its role as a master regulator.
If a mouse is genetically engineered to have no CB1 receptors, the glutamate fibers of their cerebellum get screwed up. CB1 receptors also help with brain development via their use of glutamate throughout the hippocampus and cortical layers. Scientists have confirmed that cannabidiol (CBD) affects the genetic expression of glutamate, GABA, and dopamine – but an October 2020 study shows that cannabigerol (CBG) appears to do this as well.
Scientists have also determined that CB2 cannabinoid receptors form “heteromer complexes” with a glutamate receptor known as NMDA. Heteromer complexes, a rather recent discovery, occur when two or more membrane receptors of different types entangle and combine to form a joined receptor complex that has different properties, binding actions, and downstream effects than either of the receptors on their own. At first, researchers didn’t even think CB2 receptors existed in the brain, but now we know that they’re present at low levels as guardians of brain health and that when the brain is insulted, the level of CB2 receptors shoots up to help manage the immune response. The ability of CB2 receptors to form complexes with a major glutamate receptor speaks to the therapeutic potential of cannabinoids for neuronal diseases.
A Deep Science Stroll Through the Glutamate Garden
As for the many diseases, let’s take a stroll through the glutamate garden:
Addiction
- In rats, CBD helped with the self-administration and drug seeking behavior via the ECS’s alteration of the glutamate system
- In rats addicted to cocaine, giving them anandamide (the first endocannabinoid neurotransmitter discovered) restored their glutamate homeostasis and helped to prevent cocaine relapse
- In young rats with brains damaged by adolescent alcohol intake, simply enriching their environment helped to improve their synaptic plasticity via their cannabinoid and glutamate receptors
Alzheimer’s Disease
- In a mouse model, 3 months of palmitoylethanolamide (PEA), an endocannabinoid-like compound, “rescued cognitive deficit, restrained neuroinflammation and oxidative stress, and reduced the increase in hippocampal glutamate levels”
- In a similar study, PEA improved learning, memory, and depression via rebalancing of glutamatergic transmission
- In rats with dyskinesia (involuntary erratic movements), activating the CB1 receptor helped via changes in dopamine and glutamate
Autism
- A brain imaging study shows how CBD helps via GABA and glutamate pathways
- In humans, 600 mg of cannabidivarin (CBDV – a cousin of CBD with a shorter tail) helped to modulate the GABA and glutamate systems
Depression
- The antidepressant effects of CBD in mice seem to be mediated not only by serotonin signaling but also glutamate signaling
Obesity & Metabolic Disorders
- In mice, the activation of CB1 receptors in a key brain area produces glutamate that drives the overconsumption of food
- In mice, the CB1 receptors of the brain’s hypothalamus (feeding control center) drives the response to food availability and energy balance via glutamatergic inputs
Exercise
- In mice, the ability of exercise to activate the CB1 receptor helps to enhance learning and memory via glutamate
- In mice, CB2 receptors are found on the glutamate neurons of the brain’s red nucleus (involved with motion)
Hormones
- In mice, hormones work their effects on glutamate levels via the ECS
- On the other side of the coin, the ECS regulates the hormones via glutamate in an important negative feedback loop
Multiple Sclerosis (MS)
- CB1 regulates glutamate transmission – important because excessive synaptic transmission caused by too much glutamate is a major cause of the disease’s damage
- In a mouse model of MS, those lacking CB1 receptors fared worse
Nerve Injuries
- In mice with a painful nerve injury, PEA restored cognitive behavior and neuronal functioning (long term potentiation) via two receptors in the glutamate system
- In other mice with painful nerve injuries, PEA restored their glutamate functioning and changes in amino acid release
Neuroprotection
- In microglial cells (the guardian immune cells of the brain), CBD inhibited glutamate’s increase of inflammation
- In a rat brain model of stroke, activating both the CB1 and CB2 receptor caused robust neuroprotection via lowering of glutamatergic excitotoxicity
- In neurons overexcited by glutamate, PEA helped them to calm down and to prevent neuron loss
- In early work on the antioxidant properties of the cannabinoids, CBD was found more effective than ascorbate or alpha-tocopherol against glutamate neurotoxicity
Schizophrenia & Psychosis
- In humans with psychosis, a single dose of 600 mgs of CBD increased levels of glutamate in the hippocampus and significantly decreased symptom severity
- However, in the early stages of psychosis, the heavy usage of cannabis may alter the glutamate levels and an imaging study found a relationship in first episode psychosis between the endocannabinoid system and glutamate
- Some good news: in a rat model of schizophrenia, CBD restored glutamate and GABA signaling deficits
- And ketamine seems to work well with CBD, suggesting their combined use for schizophrenia
Seizures
- Mice with febrile seizures (caused by a spike in body temperature), CBD lessened the chance of seizure via glutamate and AMPA receptors
And finally, the harms
- In adolescent rats, an IV drip of THC caused them to have less CB1, GABA and glutamate receptors in their prefrontal cortex
- In rats, prenatal exposure to a cannabinoid caused problems in their baby’s hippocampus (memory center) via dysfunction in glutamate release
- In teens who smoke heavily, it may impair the levels of glutamate in their hippocampus (memory region), though it’s hardly secret that weed isn’t great for memory – which is why it’s good for PTSD (less memories)
As always, more clinical research is needed, but for anyone with these indications or areas of concern, it’s relatively easy to start harnessing the power of glutamate and the ECS to improve your health.
See other articles in the Get to Know a Neurotransmitter series.
Lex Pelger writes articles about psychoactives and the endocannabinoid system. He publishes a weekly cannabinoid science newsletter Cannabinoids & the People and conducts 1-on-1 education sessions on using CBD, PEA, THC & CBDA for serious health conditions. © Copyright, Project CBD. May not be reprinted without permission.
Recommended Readings
Endocannabinoid Discovery Timeline
25 years of groundbreaking research — an annotated history.
Concussions & Alcoholism
Traumatic brain injury (TBI) leads to numerous problems, including alcoholism and suicide.
Ketamine Functions Through CB1
Ketamine is a dissociative anesthetic that has shown some promise for treating severe depression.